The problems don’t seem to be ending for Elon Musk. A group of doctors has called for an investigation into Neuralink, the biotechnology startup founded by the magnate, for the suspicious deaths of a group of monkeys. According to the accusations, these monkeys were allegedly used for experiments involving brain implants.
It was the Physicians Committee for Responsible Medicine (PCRM) that filed a complaint with the U.S. Securities and Exchange Commission, alleging that the primates had died due to complications with the neural implants developed by Neuralink.
Elon Musk confirmed on September 10th that a group of monkeys had died from a series of terminal illnesses. The South African entrepreneur clarified in an interview that for the initial implants, the primates used were already “near death.” He emphatically denied that the deaths were related to Neuralink’s implants. In fact, Musk stated last fall that they were being “extremely careful” with these trials.
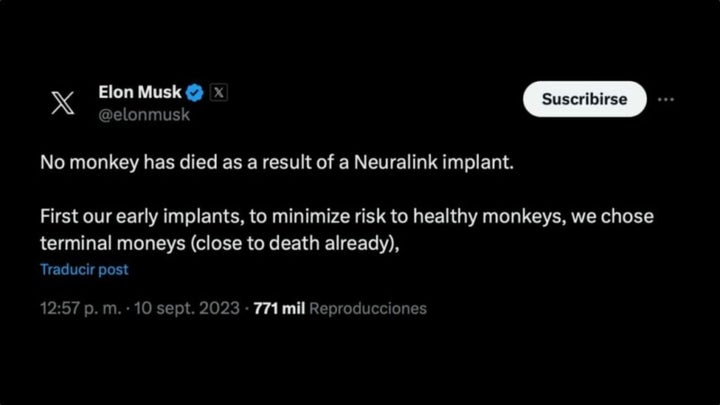
However, the group of doctors claims that these comments are “false.” The PCRM obtained a series of reports demonstrating that the monkeys had undergone “extreme suffering.” WIRED also reported on these records, confirming the animals’ horrific ordeals.
The Committee also clarifies that Musk’s statements about the monkeys’ condition do not represent the truth. While some of the animals had physical traumas and had suffered the aftermath of other experiments, “there is no evidence that they were near death.”
Neuralink is seeking volunteers to test their brain implants
Neuralink’s chips are part of the project called “Precision Robotic Implantable Brain-Computer Interface” (PRIME). These implants are designed to assist people with motor injuries or diseases such as amyotrophic lateral sclerosis (ALS).
Now, Neuralink is looking for individuals willing to participate in their first human trial. In the company’s blog, they explain that the PRIME study has received FDA approval and represents a significant step in their mission to “restore autonomy to people with medical needs.”
However, to participate, you must have tetraplegia due to cervical spinal cord injury or ALS. For more details, you can visit the website through this link.


